Letybo®
Meet Letybo®: A New Generation Wrinkle Relaxer
REQUEST A CONSULTATION
Letybo® is a next-level botulinum toxin now FDA-approved for smoothing fine lines and wrinkles with impressive precision. What sets it apart? Its advanced purification process. By removing unnecessary proteins, Letybo® helps lower the risk of developing resistance while preserving the strength needed for results that last. Keep reading to learn more.
Refined Results Without the “Frozen” Look
$12.50/unit
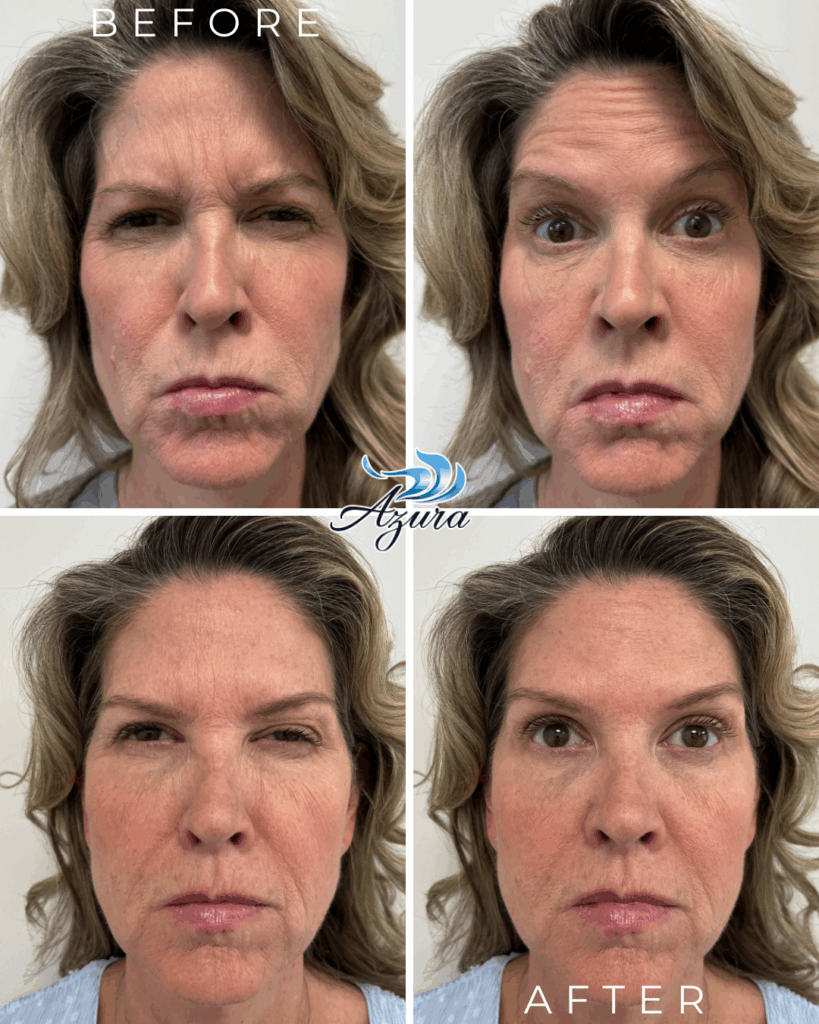
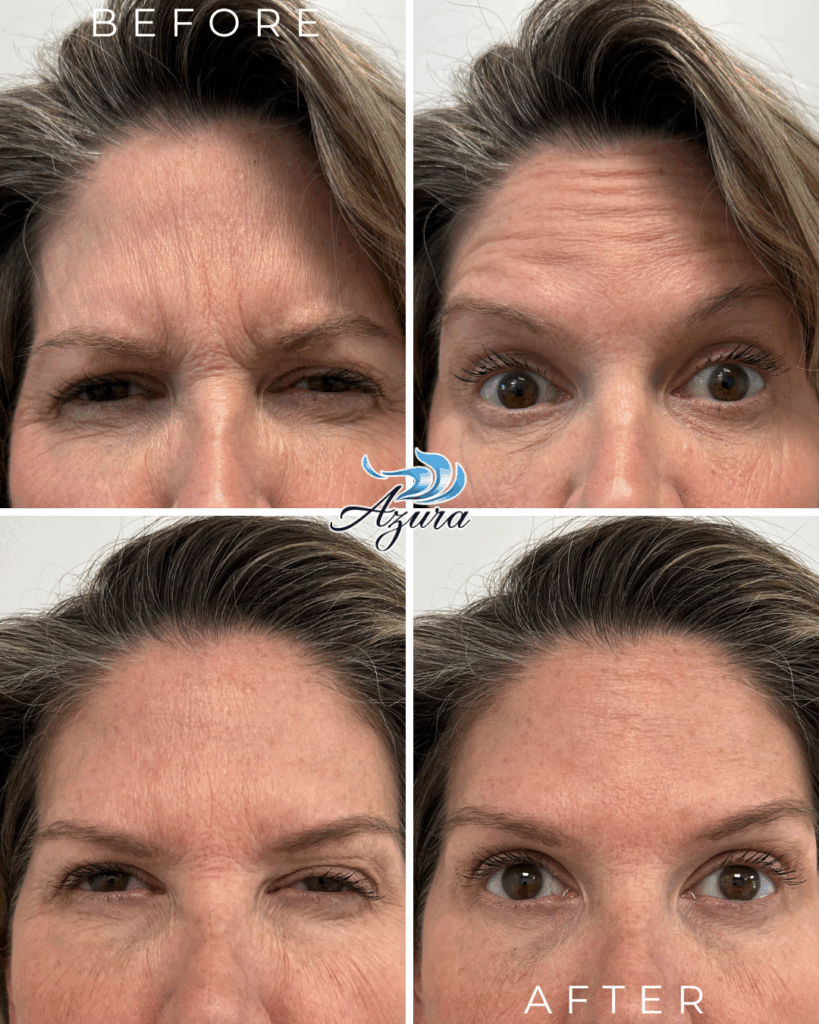
One of the top priorities for anyone exploring wrinkle relaxers is maintaining natural facial movement. Letybo® delivers smoother, more balanced results thanks to its even diffusion pattern. It’s designed to soften expression lines—like crow’s feet, forehead lines, and frown lines—while keeping your natural look intact.
There is a $50 consult fee associated with a Letybo® consultation that will go toward any purchase made or service provided at Azura.
Targeted Precision Where It Matters Most
Letybo® stands out for its ability to stay exactly where it’s placed—minimizing spread to surrounding areas. This precision makes it ideal for treating specific fine lines while keeping neighboring muscles active and expressive. If you’re looking for a more refined, natural refresh without over-relaxing your features, Letybo’s pinpoint accuracy is a major plus.
Results That Go the Distance
While every patient is different, many report results that are not only comparable to BOTOX® but may even last a bit longer. Most see smoother skin for about three to four months, with some enjoying the effects well beyond that. It’s a reliable choice for those who want longer-lasting confidence with minimal upkeep.
Important Safety Information
CONTRAINDICATIONS
Letybo contraindications include known hypersensitivity to any botulinum toxin preparation or to any of the components in the Letybo formulation and infection at the injection site.
WARNINGS AND PRECAUTIONS
Spread of toxin effects; swallowing and breathing difficulties can lead to death. Seek immediate medical attention if respiratory, speech or swallowing difficulties occur. The potency Units of Letybo are not interchangeable with other preparations of botulinum toxin products. Potential serious adverse reactions after Letybo injections for unapproved uses. If a hypersensitivity reaction occurs, discontinue Letybo and immediately initiate appropriate therapy. Adverse reports have been received involving the cardiovascular system, some with fatal outcomes. Use caution when administering to patients with pre-existing cardiovascular disease. Concomitant neuromuscular disorder may exacerbate clinical effects of treatment. Use with caution in patients with compromised respiratory function or dysphagia.
ADVERSE REACTIONS
The most common adverse reaction was headache (2%).
DRUG INTERACTIONS
Co-administration of Letybo and aminoglycosides, anticholinergic agents, or any other agents that interfere with neuromuscular transmission may potentiate the effect of Letybo; co-administer only with caution and close observation.
USE IN SPECIFIC POPULATIONS
Letybo is not recommended for use in children, pregnant or lactating women.
Please see Letybo full Prescribing Information, including Boxed Warning and Medication Guide at bit.ly/3UbRZtP